Molecular sieve adsorbents are crystalline alumino-silicates. Their unique structure allows the water of crystallization to be removed, leaving a porous crystalline structure. The corresponding crystallographic structure is formed by tetrahedras of (AlO4) and (SiO4). These tetrahedras are the basic building blocks for various zeolite structures, such as zeolites A and X, the most common commercial adsorbents.
These pores or “cages” want to readsorb water or other molecules. Aided by strong ionic forces caused by the presence of cations such as sodium, calcium and potassium, the molecular sieve will adsorb a considerable amount of water or other fluids. If the fluid to be adsorbed is a polar compound, it can be adsorbed with high loadings even at very low concentrations of the fluid.
This strong adsorptive force allows molecular sieves to remove many gas or liquid impurities to very low levels (PPM or less).
Another feature of molecular sieve adsorbents is its ability to separate gases or liquids by molecular size. The pore or “cage” openings are of the same size as many molecules. In the case of hydrocarbon paraffins, the normal, straight chained molecules can fit into the pores and be adsorbed while the branched chain molecules cannot enter the pores and pass by the molecular sieve adsorbents unabsorbed.
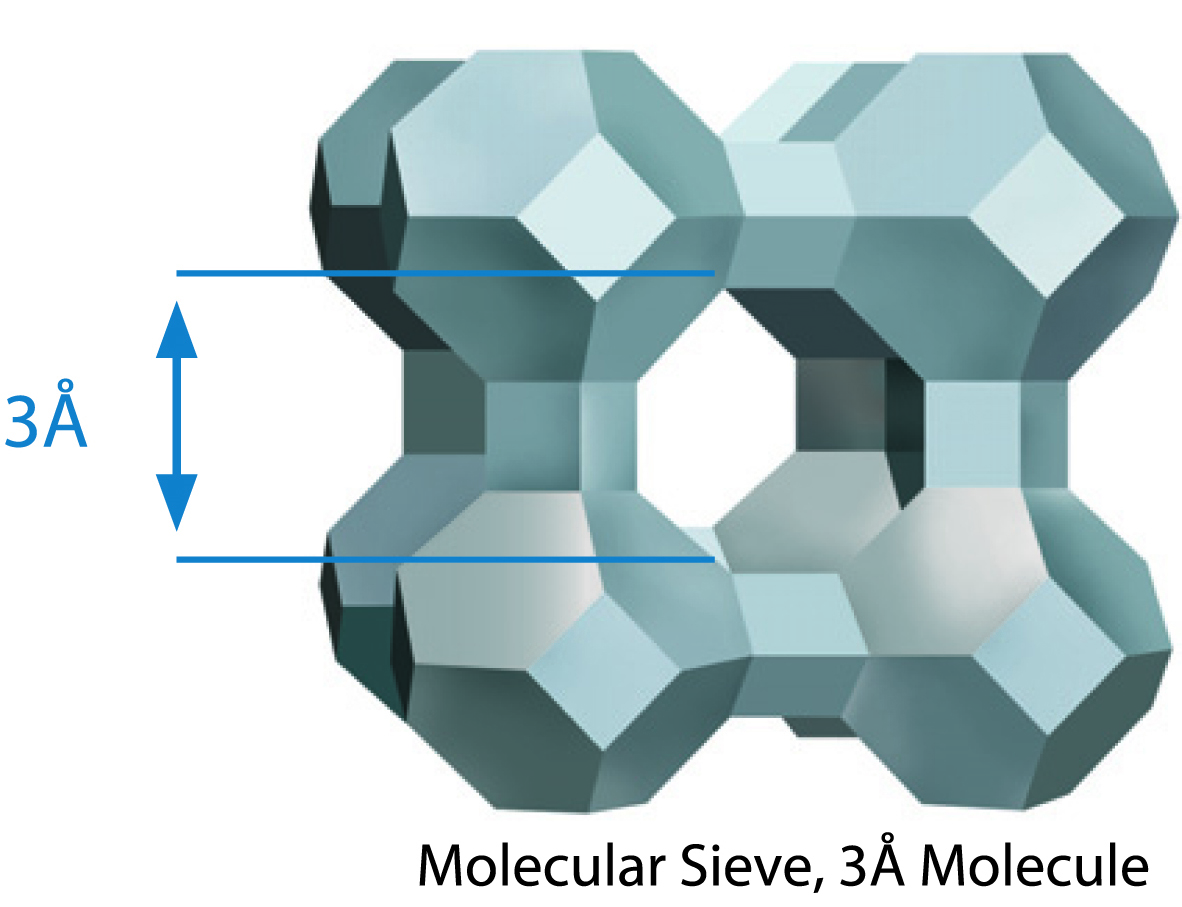


Type 3A
Molecular sieve type 3A excludes most molecules except water. It is used for natural gas dehydration, cracked gas drying, olefins drying, ethanol drying, etc. It is usually made by ion exchanging potassium onto a Type 4A in place of sodium.
Type 4A
Type 4A is a sodium form of the crystal Type and an inexpensive adsorbent usually used for drying. Further processed forms can be used for carbon dioxide removal.
Type 5A
It is the calcium exchanged form of the A crystal. The strong ionic forces of the divalent calcium ion make it an excellent adsorbent for removing carbon dioxide, hydrogen sulfide and other weakly polar molecules. This product is also used for separating normal straight chain hydrocarbons from branched hydrocarbons.
Type 13X
The sodium form of the Type X crystal has a much larger pore opening than the Type A crystals. It also has the highest theoretical capacity of the common adsorbents and very good mass transfer rates. It can remove impurities too large to fit into a Type A crystal and is commonly used to separate nitrogen from oxygen.
Molecular Sieve 13X is used commercially for general gas drying, air plant feed purification (simultaneous removal of H20 and CO2) and liquid hydrocarbon and natural gas sweetening (H2S and mercapstan removal). All molecules which can be adsorbed on molecular sieves 3A, 4A and 5A can be adsorbed on Type 13X. In addition, Type 13X can adsorb molecules of larger critical diameters, such as aromatics and branched-chain hydrocarbons.
Type Y & ZSM-5
Similar to the X crystal except it has a higher silica to alumina ratio. It is better than 13X in some applications. In the high silica/alumina ratios it can adsorb hydrophobic molecules in the presence of water and has mild acid resistance.
Activated Molecular Sieves
Activated Molecular Sieves are dehydrated fine-particle molecular sieves with high speed absorption and dispersion capability. Good additives for paint, resin, and adhesive to remove water, eliminate bubbles, improve dispersion quality, and increase strength.
Three types of activated molecular sieves are 3A, 4A, and 13X.
